2018年6月4日,博士研究生Santosh Kumar Bose和硕士研究生殷晓同学在组会上做了文献报告,具体内容如下:
Name: Santosh Kumar Bose Title: Transcriptomic Analysis in Strawberry Fruits RevealsActive Auxin Biosynthesis and Signaling in the Ripe Receptacle
Main Content: Fruit ripening is a complex and coordinatedirreversible developmental process that leads to the production of a soft andedible ripe fruit. The hormone auxin plays a critical role in fruit growth,from flower formation to fruit ripening. The auxin content of the receptacleremains constant during ripening. Two major pathways,the tryptophan dependent and -independentpathways, for IAA biosynthesis, have been reported for auxin biosynthesis in plants. However, it is accepted thata significant amount of auxin synthesis inplants is predominantly synthesized by thetryptophan-dependent two-step pathway that hasas an intermediate indol-3-pyruvic acid (IPyA). The initial conversion of Trpto IPya is catalyzed by a tryptophaneaminotransferase enzyme that includes multiple membersin several species (TAA1 and TARs in Arabidopsis). IPyA is then furtherconverted to IAA by the YUCCA proteins, a family of flavin-dependentmonooxygenases. Analysis of the transcriptome of ripening strawberryfruit revealed the changing expression pattern of the genes of auxin synthesis,perception, signaling and transport along with achene and receptacledevelopment from the green to red stage. For the synthesis of auxin, two genesencoding tryptophan aminotransferases, FaTAA1 and FaTAR2, were expressed in thered receptacle, with FaTAR2 expression peaking at this stage. The auxin activityin the ripening receptacle is supported by the DR5-directed expression of a GUSreporter gene in the ripening receptacle of DR5-GUS transgenic strawberryplants. Clustering by co-expression of members of the FaAux/IAA and FaARFfamilies identified five members whose transcriptional activity was increasedwith the onset of receptacle ripening. Among these, FaAux/IAA11 and FaARF6aappeared, by their expression level and fold-change, as the most likelycandidates for their involvement in the auxin activity in the ripening receptacle.So, it can be concluded that auxin not only plays role in development of fruitsbut also have potential effects during ripening.

Fig.1.Changes inendogenous auxin levels during receptacle development and ripening. Endogenouslevels of indole-3-acetic acid (IAA) in green, white and red receptacle asmeasured by GC-MS and expressed per gram of fresh weight (FW, A) or dry weight(DW, B). Bars represent the mean of four independent biological samples SE.Different letters indicate a significant difference between samples accordingto the corresponding ANOVA (P < 0.05).
From Fig.1, the highest IAA content (5.14 ng/g FW)was observed in the green receptacle and it was then diminished in the white (3.43ng/g FW) and red (3.18 ng/g FW) receptacle. This pattern of diminutionin the transition from green to white and red stages was altered when thevalues were expressed on a dry weight basis.
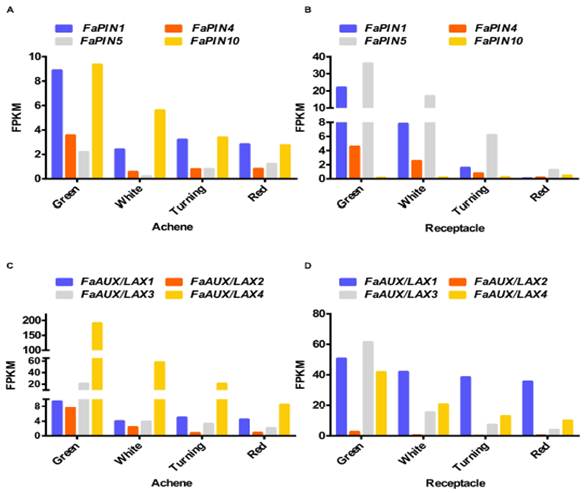
Fig.2.Expression ofthe four members of the FaPIN (A,B) and FaAUX/LAX (C,D) gene families in theachene (A,C) and the receptacle (B,D) at four developmental stages (green;white; turning; red), by RNAseq. FPKM, fragments per kilobase of exon permillion fragments mapped.
Auxin distributionby polar auxin transport is mediated by the PIN and AUX/LAX proteins. Only fourFaPIN genes (FaPIN1, FaPIN4, FaPIN5, and FaPIN10) were expressed in the fruitsof the cultivated strawberry (Fragaria ananassa), showing adevelopmental specific pattern (Figures 2A,B). While FaPIN10 had higher expressionin the achene than in the receptacle, the other three FaPIN genes displayedhigher expression in the receptacle. In all cases, the pattern corresponded todecreased expression in the transition from the green to red stage, both in theachene and the receptacle. Regarding theF.ananassaAUX/LAX genes (FaAUX/LAX1, FaAUX/LAX2, FaAUX/LAX3, andFaAUX/LAX4), all were expressed in the receptacle and achenes (Figures 2C,D).In achenes, the highest expression corresponded to FaAUX/LAX4, while the otherthree members showed higher expression in the receptacle. In all cases, as occurredfor FaPIN genes, the pattern was a continuous decrease from green to red stagesin both the achene and receptacle. It is remarkable that most of the transportgenes showed higher expression in the receptacle compared to the achene.
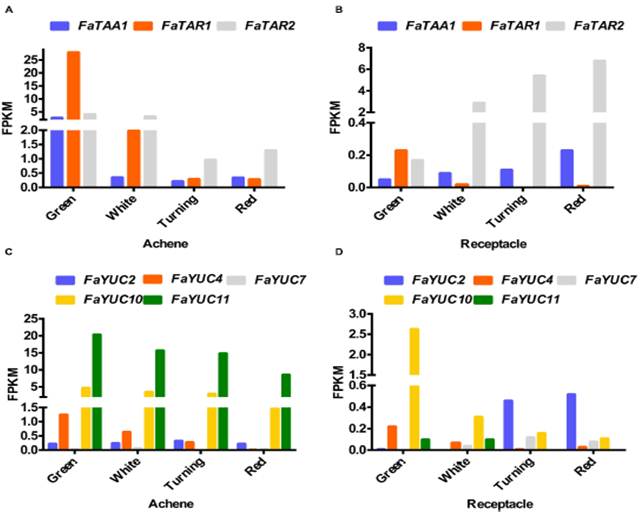
Fig.3.Expression ofthe three members of the tryptophan aminotransferase gene family (FaTAA1,FaTAR1, and FaTAR2) and four members of the flavin-dependent monooxygenases(FaYUC2, FaYUC4, FaYUC7, FaYUC10, and FaYUC11) in achene (A,C) and receptacle(B,D) at four developmental stages (green; white; turning; red), by RNAseq.FPKM, fragments per kilobase of exon per million fragments mapped.
The expression ofthe correspondingF. ananassagenes,analyzed by RNA-seq, shows that only three (FaTAA1, FaTAR1, and FaTAR2) areexpressed in fruits (Figures 3A,B). In theachene, the three genes show the same pattern,i.e., a steep decrease in the transition from the green to the white stage (Figure 3A) and then decreasingto the red stage. The highest absolute valuefor expression is shown by FaTAR1. In thereceptacle, the only gene showing expression over 1 FPKM is FaTAR2 (Figure 3B). Interestingly, theexpression of this gene continuously increasesfrom the green to the red stage. In relationto the YUCCA genes, only five of the nine genes mappedinF. vescaare expressed inF. ananassa(Figures 3C,D). In the achene, the common pattern isa decrease from green to red, with the highestexpression level being that of the FaYUC11gene (Figure 3C). Even with low expression, some YUCCA genes were identified in the red receptacle (Figure3D). In the transition from the green to the red receptacle, thereis a significant increase in the expression of both FaTAA1 and FaTAR2. Theincrease was higher for FaTAR2, which also showed expression levels that werehigher than those of FaTAA1 in the receptacle (Figure 3B).
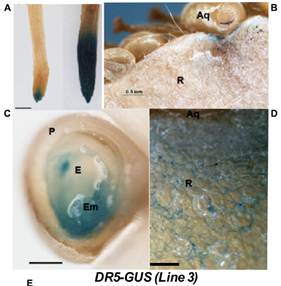
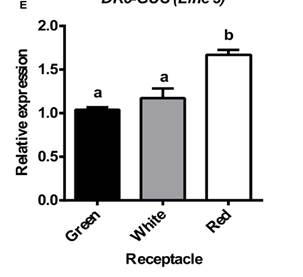
Fig.4. Auxindistribution in strawberry. DR5-GUS expression pattern in strawberry roots (A)and green fruits (B–D). For the NAA treatment, in (A), plants were grown invitro and treated with a solution of 50 mM NAA in water for 24 h (right), orjust with water for control plants (left). Scale bars(B) = 500 mm, (C,D) = 250 mm,. P, pericarp; E, endosperm; Em, embryo;Aq,achene;R, receptacle. (E) Relative expression by qRT-PCR of the GUS gene in receptacleat three developmental stages. Different letters indicate a significantdifference between samples according to the corresponding ANOVA (P < 0.05).
To investigate the role played by auxin in strawberry fruit development, thesynthetic auxin response promoter DR5 fused to the b-glucuronidase (GUS) gene wasused to generate transgenic strawberry plants. The roots of the transgenicstrawberry plants showed a GUS expression pattern that was similar to that observed in the corresponding transgenic Arabidopsis (Figure 4A),indicating that GUS expression in DR5-GUS transgenic strawberry plants is abona fide indicator of auxin output signaling. Moreover, treatment with NAAincreased the GUS staining in the root (Figure 4A), indicative of afunctional response of the DR5 promoter to auxin in this organ. In the greenfruit, DR5-driven GUS staining was observed in the receptacle around the acheneattachment (Figure 4B). A closer view of a sectioned achene showedstrong GUS staining in the embryo (Figure 4C). In the green receptaclesurrounding the achenes, we observed a gradient of GUS staining that initiatedat the achene (Figure 4D). Then, GUS transcription was evaluated inthe receptacle at three stages. The result showed that in the red receptacle, theexpression was maintained, and slightlyhigher, compared to the green and white stages(Figure 4E). Overall, the pattern of GUS activity and GUS expression in strawberry fruits confirms thepreviously reported auxin activity in theachene and green receptacle and the auxin responsivenessin red receptacle.
The pathway startswith the auxin receptor genes TIR1 and AFB. In the F. vesca genome, four auxin receptorsare present, while three are expressed in F.ananassa. Their study in the receptacle shows that FaTIR1 slightlyincreases its expression from the green to red stage, while the expression of FaAFB2decreases, and FaAFB5 remains constant (Figure 5A). Therefore, the expression in the developing receptacle of thecorresponding genes of F. ananassa (FaAux/IAA and FaARF) was analyzed in the RNA-seq expression data. A total of 19FaAux/IAA genes were expressed in ripeningreceptacle of strawberry fruit with differentpatterns (Figure 5B). Most of them showed the highest expression at thegreen stage and then continuously decreased up to the red stage, in agreement withthe occurrence of auxin in the green receptacle (Figure 5B).However,three genes showed a different pattern. The FaAux/IAA8a gene showed the highestexpression at all stages, with a small increase from the green to increase from77 FPKM at the green to 236 FPKM at the red stage (Figure 5C).Clustering by co-expression analysis of the FaAux/IAA and FaARF genes in thereceptacle during ripening was performed to identify the possible positiveinteractions between members of the two gene families (Figure 5D). Twomain clusters are formed. One of them (upper) includes those genes with the highestdecrease in the transition from the green to red stage. The other clusterincludes genes that showed variable and not very drastic changes in expressionduring the growth and ripening of the receptacle. In the lower part of thiscluster are grouped, in a sub-cluster, those genes whose expression increased with theprogression of ripening (FaAux/IAA11, FaARF6a, FaAux/IAA14b, FaAux/IAA33, andFaARF16c). In this group, the two genes showing the highest expression leveland highest change in the transition from the green to red stage are FaAux/IAA11and FaARF6a.
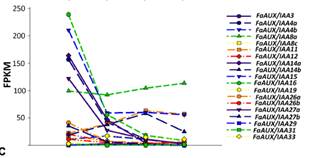
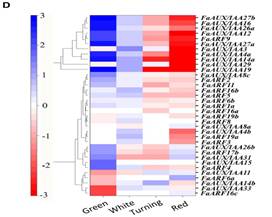
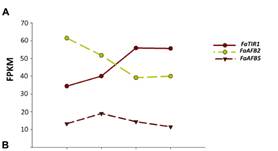
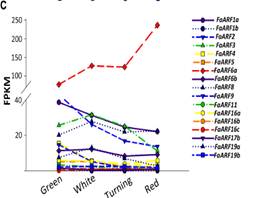
Fig.5. Expression ofthe gene family of auxin receptors (A), repressors (FaAUX/IAA (B), andtranscription factors (FaARF) (C) in the receptacle at four developmentalstages (green; white; turning; red) by RNAseq. FPKM, fragments per kilobase ofexon per million fragments mapped. (D) K-means clusters of 35 genes showingdistinct stage- and tissue-specific expression patterns. The scale: averagedlog2 “relative RPKM value” of all genes in each cluster.
The results, shown in Figure 6, confirm the RNA-seq data, i.e., there is an increase in theexpression of the genes FaAux/IAA11 (Figure 6A) and FaARF6a, (Figure 6B),jointly with the auxin receptor FaTIR1 (Figure 6C), parallel to theripening process. Altogether, these results point to the operation of the auxinresponsive components of the strawberry receptacle at the ripe stage.
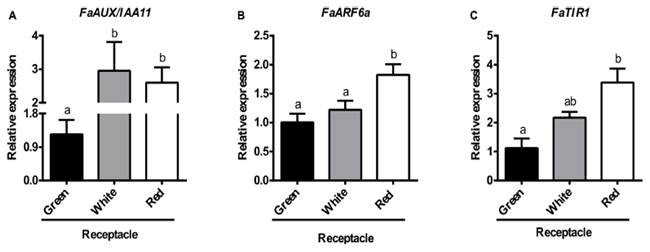
Fig.6. Relativetranscript levels of FaAUX/IAA11 (A), FaARF6a (B), and FaTIR (C), as determinedby qRT-PCR in different developmental stages of receptacle. Bars represent themean of three independent biological samples SE. Different letters indicate asignificant difference between samples according to the corresponding ANOVA (P< 0.05).
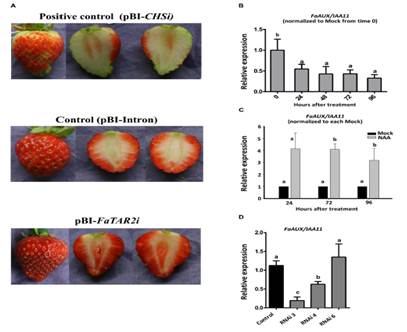
Fig.7. (A) Strawberrytransgenic fruits agroinfiltrated with empty vector (control), with theFaTAR2-RNAi construct (RNAi) and with Chalcona synthase-RNAi, FaCHS-RNAi, as apositive control at 7 days after injection. (B) FaAUX/IAA11 expression byqRT-PCR of de-achened green strawberry fruits and covered with a lanolinepaste. (C) Time course expression of FaAUX/IAA11, by qRT-PCR, of the receptacleof strawberry fruits injected, at the turning stage, with a water solution ofNAA and the corresponding control (mock solution). (D) FaAUX/IAA11 expressionin silenced fruit agroinfiltrated with empty vector (control) and withFaTAR2-RNAi construct. Error bars indicate +SE of three biological replicates.Different letters indicate a significant difference between samples accordingto the corresponding ANOVA (P < 0.05).
报告人:殷晓 报告题目:Designing hydrogels for controlleddrug delivery
报告内容:由于传统的药物治疗体系存在靶向性低,用药量大,且需重复给药的缺点,文章主要对水凝胶药物递送体系进行介绍。
1.因为水凝胶的宏观结构决定载药体系作用于人体的运输途径,文章首先对水凝胶的宏观尺寸进行介绍。
2.因为水凝胶的网格会调节药物在凝胶内部的扩散,文章接着介绍水凝胶的网孔大小对药物扩散影响机理。
3.由于水凝胶网络中分子及原子会与药物分子产生相互作用,文章对其具体结合机制进行介绍。
4.最后文章对不同水凝胶体系的释放动力学进行比较。

网孔尺寸调节药物扩散
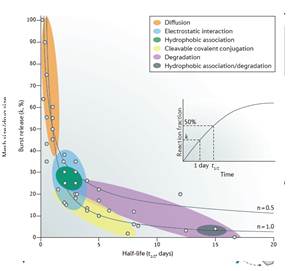
不同凝胶体系释放动力学比较